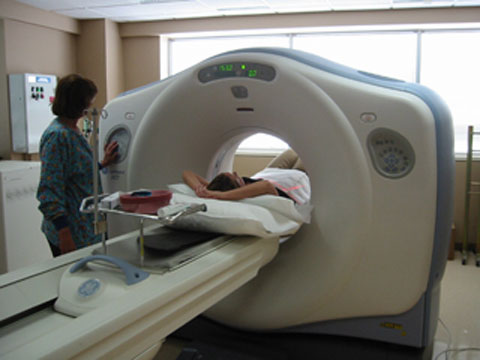
 The US Food and Drug Administration (FDA) has mentioned that manufacturers of CT scanners should focus and improve the training and learning offered to those who use their tools and that the machines themselves should be made more safer by warning workers that a harmful high radiation dose is about to be administered. These two conclusions were reported in documents prepared based on the finding of a yearlong investigation by the Food and Drug Administration about hundreds of radiation overdose cases. These cases took place in 2008 and 2009; they included cases that happened in a specific detecting exam for a stroke, named a CT brain perfusion scan.
The US Food and Drug Administration (FDA) has mentioned that manufacturers of CT scanners should focus and improve the training and learning offered to those who use their tools and that the machines themselves should be made more safer by warning workers that a harmful high radiation dose is about to be administered. These two conclusions were reported in documents prepared based on the finding of a yearlong investigation by the Food and Drug Administration about hundreds of radiation overdose cases. These cases took place in 2008 and 2009; they included cases that happened in a specific detecting exam for a stroke, named a CT brain perfusion scan.
Dr. Alberto Gutierrez, an FDA official who oversees detecting devices, explained that the full extent of the overdoses might never be identified due to controlling how and when radiation overdoses should be mentioned. He also reported “We actually don’t have a good description of what is a reportable event in radiation. There is some need for the agency to clarify what is reportable and for user facilities to have a system by which they can track and report incidents to the agency and to the manufacturer.”
Especially, Dr. Gutierrez named Huntsville Hospital in Alabama for its “unsatisfactory” accepted to brain scan overdoses there. The FDA noted that it should not certainly mention how many radiation overdoses happened at the hospital. The spokesman of this hospital, Burr Ingram, explained that his facility took any recommendation from the FDA and that it was already working to make its reporting in the same way with the agency’s guidelines.
GE Healthcare had noted that the hospital used high levels of radiation in order to provide high detailed images. Previously, the New York Times mentioned that Huntsville had some of the largest brain scan overdoses and the organization that supplied the scanners.The FDA had forwarded a letter to the Medical Imaging Technology Alliance, mentioning its recommendations, involving one that asked manufacturers to do a better job organizing dose-related information in their user manuals. The agency reported that it aimed to debate its recommendations with manufacturers at the end of this month, and that it expected the organizations to work on at least some of them.
Dave Fisher, The executive director of the manufacturers’ group, reported that his company over the last year had been acting with the F.D.A on these objects. Mr. Fisher explained that “In the end, the optimal solution will require action by all stakeholders including, manufacturers, users and regulators”.