An echocardiographic and fluoroscopic-guided catheter-based procedure is equal to conventional open-heart surgery, a new study shows, at least in terms of survival for replacement of aortic heart valves in elderly high-risk patients.
 This is the first report from The PARTNER Trial, co-led by Dr. Craig R. Smith, chief of the Division of Cardiothoracic Surgery, New York-Presbyterian Hospital/Columbia University Medical Center, who presented the findings at the American College of Cardiology’s American College of Cardiology’s 60th Annual Scientific Session. ACC.11. The PARTNER trial is designed for patients with severe calcific aortic stenosis who might not be optimal candidates for conventional open-chest valve replacement due to the risks of surgery.
This is the first report from The PARTNER Trial, co-led by Dr. Craig R. Smith, chief of the Division of Cardiothoracic Surgery, New York-Presbyterian Hospital/Columbia University Medical Center, who presented the findings at the American College of Cardiology’s American College of Cardiology’s 60th Annual Scientific Session. ACC.11. The PARTNER trial is designed for patients with severe calcific aortic stenosis who might not be optimal candidates for conventional open-chest valve replacement due to the risks of surgery.
“These results clearly show that TAVR is an excellent alternative to surgical AVR (aortic valve replacement) in high-risk patients,” Smith said in a prepared statement. “Recommendations to individual patients will need to weigh the appeal of avoiding open-heart surgery, with its known risks, against less invasive TAVR with different and less well understood risks, as well as the absence of long term follow-up. Future trials will help delineate the role of TAVR in intermediate risk patients.”
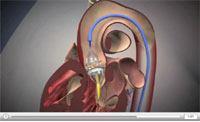
In both procedures an artificial valve is placed in the aortic valve between the left ventricle and the aorta, the main artery supplying the body with oxygenated blood. With transcatheter aortic valve replacement, or TAVR, a catheter is threaded through the femoral artery to the aortic valve using echocardiographic and fluoroscopic imaging. The collapsed valve is then pushed through the catheter and placed within the natural valve and expanded with a balloon to open the collapsed valve. A wire mesh stent on the outside of the artificial valve locks the implanted valve into place.
The large study involved 699 high-risk older patients averaging 84.1 years with severe stenosis of the aortic valve, defined as an opening of less than 0.8 centimeters squared. Patients were randomly assigned to surgery or TAVR at 26 clinical trial sites nationwide. A total of 350 patients underwent open surgery and 349 underwent TAVR.
Early study results showed that 6.5 percent of patients in the conventional surgery group died from all causes within the first 30 days after the surgery compared to 3.4 percent of the TAVR group. All-cause mortality at 1 year was similar for the two groups, at 26.4 percent for the surgery group and 22.2 percent for the transfemoral TAVR subgroup.
The rate of major strokes among patients in the surgery group, however, was about half that of the rate in the TAVR group. A total of 2.1 percent of the surgery patients suffered strokes compared to 3.8 percent of the TAVR group. Similarly, the rate of major vascular complications within the first 30 days was much lower in the surgery group at 3.2 percent, compared to 11.0 percent of the TAVR. Para-valvular regurgitation, or leaks around the replacement valve also occurred more often with TAVR. Patients in the surgery group had more bleeding complications with rates at 19.5 percent compared to 9.3 percent of the TAVR group.
Smith acknowledged the higher rates of stroke and complications in the TAVR group, but noted that the stroke rate had declined 20 percent to 30 percent in this study compared to a previous study. He expects those rates to continue to decline with improved devices and greater experience in performing the procedure. The transcatheter aortic valve replacement procedure has been performed in Europe since 1997, where more than 5,000 people have been treated.
Edwards LifeSciences, which makes the collapsible replacement valve, funded the PARTNER study. Dr. Smith has no financial relationship with the company. A second trial, PARTNER II was approved in February to test a new generation valve and delivery system against the current valve and delivery system used in this PARTNER trial.
By Michael O’Leary, contributing writer Health Imaging Hub
